Modelling extracellular matrix of human adult brain injury for repair
Meriam Ernez — Hector RCD Magdalena Götz
Brain injury and neurodegeneration cause irreversible neuron loss. This project models the adult human brain injury environment by decellularizing adult human brain tissue from patients and characterizing its extracellular matrix (ECM). Neuronal replacement strategies will be tested, including transplantation of human iPSC-derived neurons and reprogramming of iPSC-derived or adult astrocytes isolated from resection material, to define conditions that support neuronal replacement across diseases and ages.
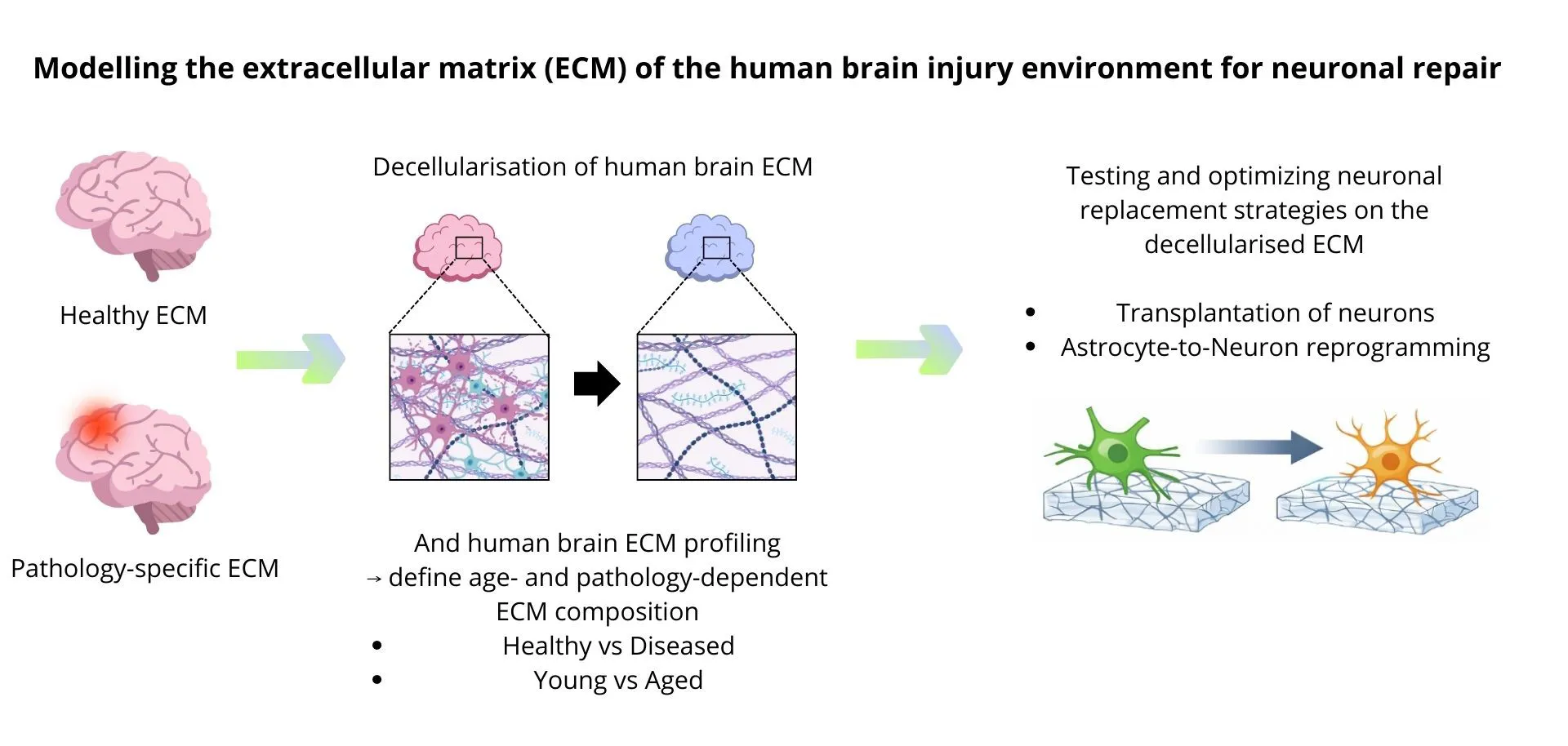
Neuronal loss occurs due to aging, neurodegenerative diseases, or stroke and is largely irreversible in humans. Regeneration in the adult human brain is very limited. Although neuronal transplantation and direct glia-to-neuron reprogramming represent promising neuronal replacement strategies, their success is strongly influenced by the injury microenvironment. After injury or disease, the extracellular matrix (ECM) changes significantly and can strongly limit neuronal survival and repair. This human injury environment remains poorly understood, which limits the development and clinical translation of regenerative strategies.
Models of adult human brain injury microenvironments are established by decellularizing postmortem brain tissue from different pathologies and age groups and characterizing ECM composition mainly using proteomics. Healthy brain ECM is compared with pathology-specific ECM to define age- and injury-associated changes relevant for regeneration. Two strategies for neuronal repair are examined: transplantation of human iPSC-derived neurons and astrocyte-to-neuron reprogramming on decellularized ECM using either young iPSC-derived astrocytes or adult astrocytes isolated from resection material. The influence of the ECM on neuronal survival, stress responses, synaptic integration, and fate conversion is systematically analyzed to define how human matrix composition affects regenerative potential and to optimize neuronal replacement strategies.
The project aims to model the human brain injury extracellular matrix (ECM) to determine how matrix composition affects neuronal repair. Replacement strategies for lost neurons are evaluated on decellularized ECM to identify ECM features that regulate neuronal survival and integration.

Meriam Ernez
Biomedizinisches Zentrum der Ludwig-Maximilians Universität, Institut für Stammzellforschung am Helmholtz Zentrum München & Max-Planck-Institut für Biochemie
Supervised by

Magdalena Götz
Medicine & BiologyHector Fellow since 2023