Inferences of clonal dynamics in human hematopoiesis
Lena Nitsch – Hector RCD Awardee Leif Ludwig
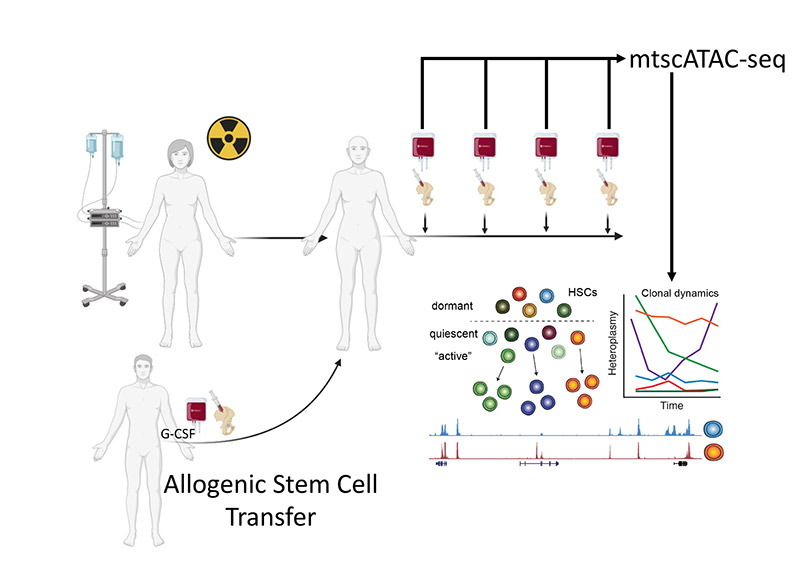
The formation of cells in our blood is sustained throughout our lifetime by hematopoietic stem cells. An understanding of the output and activity of individual stem cells, however, is only emerging. In my project, I will investigate clonal reconstitution dynamics and evaluate hematopoietic regeneration utilizing somatic mutations in the mitochondrial genome as natural barcodes in matching bone marrow and peripheral blood samples from patients undergoing allogenic stem cell transplantation.
Somatic mutations accumulate in cells throughout our lifetime and can thereby represent an accurate molecular clock. Notably, this makes them ideal for understanding developmental and regenerative processes of human physiology directly in vivo.
The hematopoietic system is an essential organ system with long-term stable contribution to all lineages of blood cells. The formation of these individual cellular parts of the blood, a process referred to as hematopoiesis, is sustained by hematopoietic stem and progenitor cells (HSPCs) which reside mainly in the bone marrow. An understanding of their lifetime dynamics and direct output of individual stem cells in humans is, however, only emerging.
An elegant way to assess the activity and output of one individual stem cell and its clonal behavior in humans in vivo is clonal tracing by using somatic variation within the mitochondrial genome. The mitochondrial genome is small enough for cost-efficient sequencing but large enough to accumulate substantial genetic diversity. Given its higher mutation rate compared to the nuclear genome, we can utilize the resulting variation as natural barcodes. Specifically, by leveraging mitochondrial DNA mutations that arise in single cell stem cells and following these mutations in longitudinally sampled, matching blood and bone marrow samples after allogenic stem cell transplantation, we envision to investigate clonal reconstitution dynamics of stem cells to evaluate hematopoietic regeneration and stem cell behavior in vivo, monitor residual disease, and characterize properties of malignant cells in case of relapse.

Lena Nitsch
Max-Delbrück-Centrum für Molekulare MedizinSupervised by

Leif S. Ludwig
Biology & Medicine
